This article has been written in response to the upcoming Rare Disease Day 2023. Rare Disease Day is raising awareness and generating change for the 300 million people worldwide living with a rare disease, their families and carers. For more information, visit rarediseaseday.org.
Rare diseases, also known as orphan diseases, are defined as conditions that affect fewer than 200,000 people in the United States or less than 5 in 10,000 individuals in Europe. Despite their rarity, orphan diseases affect an estimated 300 million people worldwide, and around 95% of rare diseases have no approved treatments. The development of mRNA-therapies for rare diseases has been gaining attention as a promising new frontier for addressing the unmet needs of patients with rare diseases.
mRNA-technology has the potential to treat various diseases, including rare ones, due to its precise and customized treatment capabilities. mRNA-therapies offer several advantages, including fast development and manufacturing. This makes them ideal for treating rare diseases with limited treatment options. Additionally, this therapy can be personalized to each patient based on their specific genetic makeup, enabling the creation of individualized treatment plans.
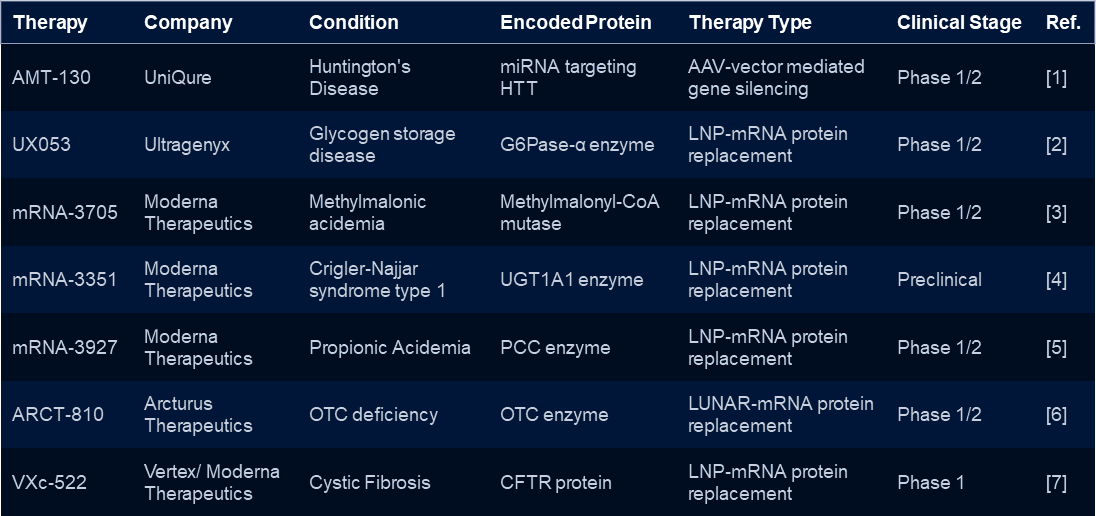
Table 1: mRNA-based therapies for rare diseases: Please note that this table may not be exhaustive and the information may be subject to change as clinical trials progress.
Note: CFTR refers to the cystic fibrosis transmembrane conductance regulator protein, UGT1A1 refers to UDP-glucuronosyltransferase 1A1, PCC refers to propionyl-CoA carboxylase, OTC refers to ornithine transcarbamylase, and LUNAR refers to lipid-enabled and unlocked nucleic acid modified RNA delivery technology.
Cystic Fibrosis: VXc-522
Vertex Pharmaceuticals’ mRNA-therapy for cystic fibrosis, known as VX-880, is an example of the potential of mRNA-therapies to treat rare diseases. Cystic fibrosis is a rare genetic disorder that affects the lungs and digestive system. Currently, there is no cure for the disease. VX-880 is designed to provide cells with the instructions to produce a functional cystic fibrosis transmembrane conductance regulator (CFTR) protein. This protein is missing or defective in people with cystic fibrosis. The therapy is currently in phase 1/2 clinical trials, and shows promise in improving lung function in patients.
Crigler-Najjar syndrome: mRNA-3351
Moderna Therapeutics is also actively developing mRNA-therapies for rare diseases, including therapies for methylmalonic acidemia, Crigler-Najjar syndrome, and propionic acidemia. The Crigler-Najjar syndrome is a rare inherited disorder that causes a build-up of bilirubin in the blood. This can lead to severe jaundice and potentially life-threatening complications. Moderna’s mRNA-therapy for Crigler-Najjar syndrome, known as mRNA-3351, is designed to provide cells with the instructions to produce a functional enzyme that can break down bilirubin in the blood. The therapy is currently in preclinical development, and Moderna is planning to initiate clinical trials in the near future.
Methylmalonic acidemia: mRNA-3705
Methylmalonic acidemia and propionic acidemia are rare genetic disorders that affect the body’s ability to break down certain amino acids and fats. This leads to a build-up of toxic substances in the body. Moderna’s mRNA-therapy for methylmalonic acidemia, known as mRNA-3704, is designed to provide cells with the instructions to produce a functional methylmalonic CoA mutase (MUT enzyme), which is missing or defective in people with the disease. The therapy is currently in phase 1/2 clinical trials, and the preliminary results have shown promise in reducing levels of toxic substances in the blood.
OTC deficiency: ARCT-810
Arcturus Therapeutics is also developing an mRNA-therapy for ornithine transcarbamylase (OTC) deficiency. OTC deficiency is a rare genetic disorder that affects the liver’s ability to break down nitrogen, leading to a build-up of toxic ammonia in the body. ARCT-810 works by using mRNA to deliver a functional OTC gene to liver cells. There, it can produce the missing OTC enzyme and improve the body’s ability to process ammonia.
Preclinical studies of ARCT-810 have shown promising results, with significant reductions in ammonia levels and improvements in survival rates in animal models. Currently, Arcturus is conducting a Phase 1/2 clinical trial to assess the safety and efficacy of ARCT-810 in human patients with OTC deficiency. If successful, this treatment could offer a new hope for patients with this rare and potentially life-threatening condition.
Conclusion
mRNA-therapies show great potential in treating rare diseases with genetic origins due to their specificity, reduced off-target effects, and favorable safety profile. The success of current therapies has spurred the development of more treatments. However, challenges still exist, such as the need for improved delivery systems, the cost of treatment, and the difficulty of clinical trial recruitment due to the rarity of the diseases.
Despite these challenges, the future of mRNA-therapies for rare diseases looks promising. As more research is conducted and clinical trials are completed, the potential of mRNA-therapies to treat rare diseases will become increasingly evident. The table provided in this article highlights some of the most promising mRNA-therapies for rare diseases in development, giving hope to patients and families affected by these devastating conditions.
References:
[1] ClinicalTrials.gov Identifier: NCT05243017
[2] ClinicalTrials.gov Identifier: NCT04990388
[3] ClinicalTrials.gov Identifier: NCT05295433
[4] Program Update (04/May/2022): Moderna’s therapeutics: Crigler-Najjar Syndrome Type 1 (CN-1)
[5] ClinicalTrials.gov Identifier: NCT04159103
[6] ClinicalTrials.gov Identifier: NCT04442347
[7] ClinicalTrials.gov Identifier: NCT05668741